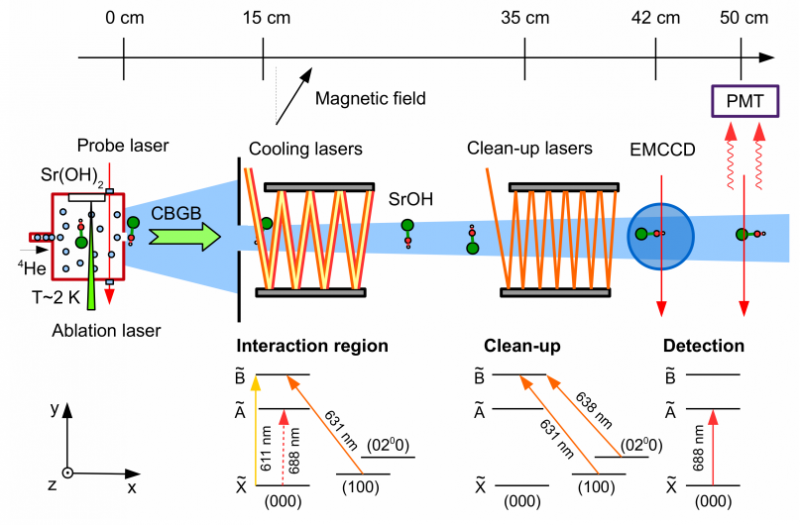
Schematic of the experimental apparatus (not to scale). Credit: arXiv:1609.02254 [physics.atom-ph]
(Phys.org)—A team of researchers at Harvard University has successfully cooled a three-atom molecule down to near absolute zero for the first time. In their paper published in Physical Review Letters, the team describes how they achieved the feat and suggest that their technique could be modified to allow for cooling molecules with even more atoms.
For many years, scientists have been laser cooling atoms down to near absolute zero as part of research into understanding how atoms work—the cooler temperatures slow things down, allowing for a better look. In such work, atoms are cooled due to scattering of photons, which serves to transfer momentum—electron bonds are forced to release the photons, causing the atoms to nearly cease moving. Doing the same has been harder for molecules because of their more complicated structure, i.e., their vibrational and rotational degrees of freedom.
A specific type of laser cooling of molecules called Sisyphus cooling involves creating a wave of laser light that causes the molecule to emit into a magnetic state without interaction with the laser—another smaller magnetic field is then used to bring the molecule back to its initial state. The process repeats with each step causing a loss of kinetic energy which causes the molecule to grow cooler and cooler. In this new effort, the researchers used this technique (magnetically assisted Sisyphus laser cooling) to cool a molecule with three atoms down to very near absolute zero.
The molecule (strontium monohydroxide—SrOH) was chosen due to its unique properties—it contains an electron that does not participate very strongly in bonding—which the team notes, made it an ideal candidate. It also suggests, the team further notes, that other molecules with like properties could work, as well—even some with more atoms. They suggest the technique might work with molecules that have as many as 15 atoms, and it could also be used as part of the basis for a quantum computer because it allows for changing a molecular state with precision. It might also prove useful in chemistry, as well, they note, because it could cause reactions to slow down, allowing for better observation, giving a far better level of detail.
© 2017 Phys.org