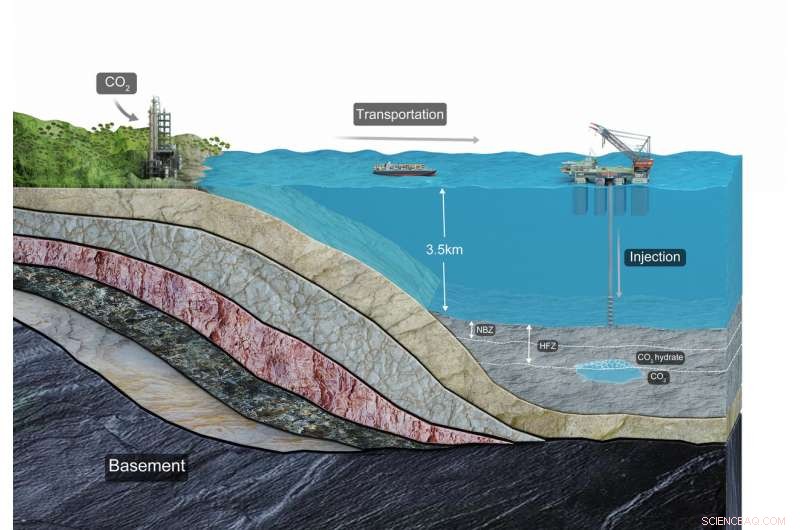
Schematic illustration of the infrastructure and related processes of carbon sequestration in deep-sea sediments. Credit: Yihua Teng and Dongxiao Zhang
A pair of researchers at Peking University has found evidence that suggests liquid CO2 could be safely sequestered in deep sea sediments. In their paper posted on the open access site Science Advances, Yihua Teng and Dongxiao Zhang describe a model they built to mimic CO2 injections beneath the ocean floor and what it showed.
As the planet continues to heat up due to the continued release of greenhouse gases into the atmosphere, scientists look for other places to store them. Carbon dioxide has been singled out as one of the major greenhouse gases and because of that, efforts have been made to curb its release. Some approaches have focused on looking for ways to prevent is release, while others look for ways to capture and store it where it will not eventually leak into the atmosphere. One such place is in sediments that lie at the bottom of the ocean. But, as the authors note, little work has been done to find out if such a site might be able to hold CO2 without leakage into the water—and eventually into the atmosphere. In this new effort, the researchers built a model meant to mimic ocean floor sediment conditions and what might happen if liquid CO2 were injected into it.
One of the major culprits involved in releasing CO2 into the atmosphere is coal-burning power plants. Work is currently being done to find ways to sequester the CO2 in these emissions. Such work has shown that CO2 can be captured and converted to various forms, from solids to liquids. It is the liquid form that the researchers with this new effort address.
Prior research has shown that when liquid CO2 is exposed to both high pressure and low temperatures, hydrates form. The researchers added this information to their model and then ran it multiple times under different conditions such as varying pressure and time scales. They found that under certain conditions, injecting CO2 into the sediments led to the formation of hydrates, which then served as a form of cap, preventing the CO2 liquid from seeping upward. They further found that over time, both the CO2 and the hydrates dissolved into pore fluids.
Emboldened by their results, the researchers suggest real-world studies of CO2 sequestration in seafloor sediments to determine if it is a viable solution.
© 2018 Phys.org