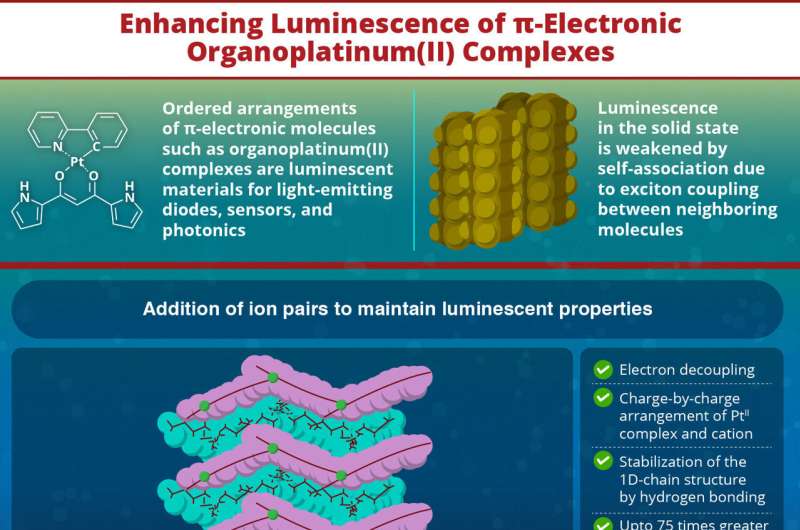
Photoluminescent molecules, capable of absorbing and re-emitting light, play an important role in the development of technologies such as light-emitting diodes, sensors, and displays. Among them, ordered arrangements of π-electronic molecules such as crystals of organoplatinum(II) complexes, where a platinum(II) ion is coordinated by organic ligands in a square-planar arrangement, stand out for their applications in energy-efficient flexible displays.
However, their luminescence in the solid state is short-lived due to the interaction between excitons (bound electron-hole pairs) of neighboring molecules. To address this issue, bulky foreign molecules are introduced into the molecular structure to prevent or minimize the electronic interactions between molecules.
Using this strategy, a research team led by Professor Hiromitsu Maeda from Ritsumeikan University, Japan, recently enhanced the solid-state phosphorescence in multiple organoplatinum(II) complexes, increasing the phosphorescence by up to 75 times.
"Spatially and electronically isolated ordered arrangement of emissive π-electronic molecules is a principal point for the preparation of emissive solid-state materials. This concept can be used in materials for organic electronics, particularly organic light-emitting diodes for flexible displays," explains Prof. Maeda.
In their study published in Chemical Science on December 5, 2023, the research team synthesized dipyrrolyldiketone PtII complexes consisting of four different C^N ligands. These molecules display strong phosphorescence in solution but show extremely weak phosphorescence in the solid state due to self-association.
To enhance their luminosity in the solid state, the team introduced ion pairs consisting of a chloride anion and tetraalkylammonium countercations: TPA+ (tetrapropylammonium), TBA+ (tetrabutylammonium), and TPeA+ (tetrapentylammonium). This resulted in ion-pairing assemblies consisting of chloride ion-binding PtII complexes and countercations.
The chloride ions bind to the PtII complex via hydrogen bonds, while the cations form layers between the π-electronic molecules. X-ray analysis confirmed the complex's rigid structure, where PtII complexes are separated by cations in charge-by-charge arrangements.
By isolating the π-electronic molecules from each other, the researchers enhanced the luminescent properties of the organoplatinum(II) complexes in the solid state. Compared to the original anion-free states where the complex is not bonded to the chloride ion, the relative intensity of phosphorescence in Cl−-binding PtII complexes with cations showed improvements ranging from 1% to 7.5%, a 75-fold increase over the original molecule.
The luminescence also lasts significantly longer, with certain ion-pairing assemblies achieving an emission lifetime nearly 200 times longer than the monomeric PtII complex. Theoretical studies using DFT calculations revealed that the charge-by-charge packing structure prevents the delocalization of the electron wavefunction over PtII complexes.
"To the best of our knowledge, such a room-temperature phosphorescence enhancement by anion binding and ion-pairing assembly has not been demonstrated thus far," remarks Prof. Maeda.
Such a strategy can be used to design emissive materials and improve the phosphorescence of solid-state materials for novel applications.
"The chemistry of ion-pairing assembly of charged π-electronic molecules is a new topic in a research area of supramolecular chemistry. Understanding the interactions between charged species and the formation of assembled structures through research will affect in a further design and fabrication of functional ion-pairing assemblies such as efficient electric conductive materials, ferroelectric materials, and chiral transfer in ion pair and the ion-pairing assemblies exhibiting fascinating optical properties," concludes Prof. Maeda.
More information: Yohei Haketa et al, Enhanced solid-state phosphorescence of organoplatinum π-systems by ion-pairing assembly, Chemical Science (2023). DOI: 10.1039/D3SC04564A
Provided by Ritsumeikan University