A recent study, published in Applied Clay Science, could have applications in clay column chromatography for obtaining enantiomeric compounds in industries.
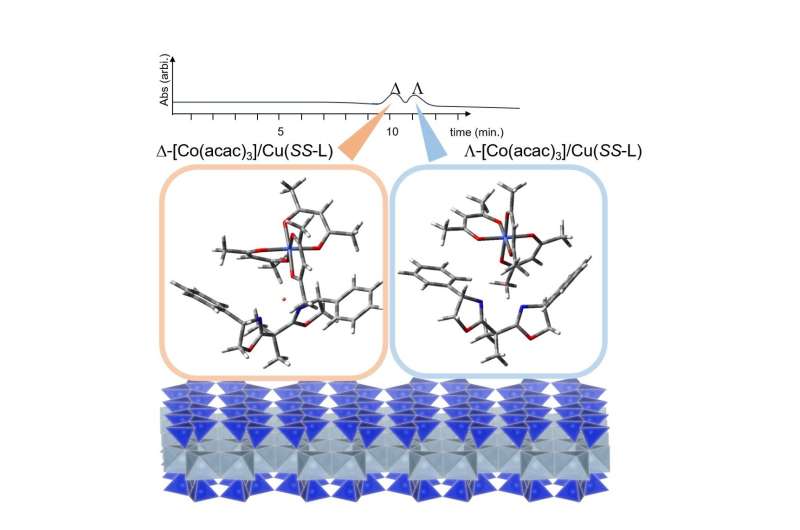
A spherically-shaped particle of synthetic hectorite (denoted as Na-HEC) was ion-exchanged with a divalent Cu(II) complex, [Cu(SS-oxa)]2+ (SS-oxa = SS-2,2′-isopropylidene-bis(4-phenyl-2-oxazoline)). The material is denoted as [Cu(SS-oxa)]2+/HEC.
A column for high performance liquid chromatography (HPLC) was prepared by packing 4.0 g of [Cu(SS-oxa)]2+/HEC into a stainless tube (25 cm x 0.4 cm (i.d.)). When tris(acetylacetonato)cobalt(III) (denoted as [Co(acac)3]) was eluted by methanol at the flow rate of 0.2 mLmin-1at 4°C, the compound was separated to D- and L-enantiomers nearly to the baseline.
Useful organic molecules with two hydroxyl groups such as 1,1'-binaphthyl-2,2'-diol were also partially resolved.
With the help of theoretical simulation, it was concluded that the resolution was realized achieved by the occupation of the enantiomers in a cavity around a Cu(II) ion. The column also exhibited resolution ability toward an organic molecule with two hydroxyl groups indicating that the molecule binds with a Cu(II) ion in a stereoselective way through coordinating interactions.
More information: Akihiko Yamagishi et al, Use of an ion-exchange adduct of synthetic hectorite and chiral copper(II) complex as a packing material for chromatographic resolution, Applied Clay Science (2024). DOI: 10.1016/j.clay.2024.107290
Provided by Ehime University