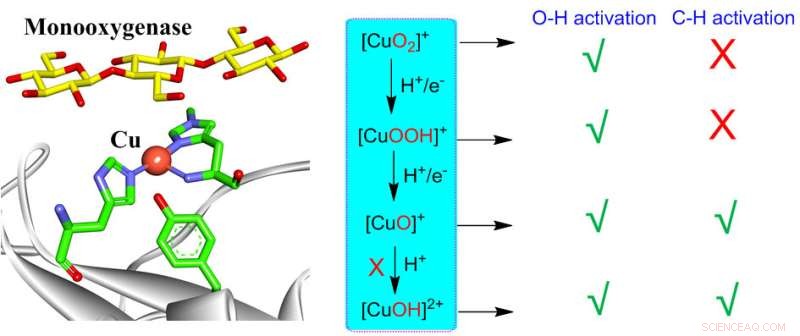
Dioxygen activation by mononuclear copper in biological and synthetic systems may generate various copper-oxygen intermediates including [CuO2]+, [CuOOH]+, [CuO]+, [CuOH]2+. All these species are able to perform O-H activation, while only [CuO]+ and [CuOH]2+ are reactive for C-H activation. However, the formation of [CuOH]2+ is highly unfavorable in monooxygenases, leaving [CuO]+ as the only active intermediate responsible for C-H activation in monooxygenases. These insights can provide consistent understanding on reactivities of various copper-oxygen active species in biological and synthetic systems. Credit: Chinese Journal of Catalysis
Dioxygen activations constitute one of the core issues in copper-dependent metalloenzymes. Upon O2 activation, copper-dependent metalloenzymes, including particulate methane monooxygenases (pMMOs), lytic polysaccharide monooxygenases (LPMOs) and binuclear copper enzymes PHM and DBM, are able to perform challenging C-H/O-H bond activations.
Meanwhile, copper-oxygen core-containing complexes have been synthetized to mimic the active species of metalloenzymes. Dioxygen activation by mononuclear copper active site may generate copper-oxygen intermediates, including Cu(II)-superoxo, Cu(II)-hydroperoxo, Cu(II)-oxyl as well as the Cu(III)-hydroxide species.
Intriguingly, all these species have been invoked as the potential active intermediates for C-H/O-H activations in either biological or synthetic systems. Due to the poor understanding of the reactivities of the copper-oxygen complex, the nature of active species in both biological and synthetic systems are highly controversial.
Recently, a research team led by Prof. Binju Wang from Xiamen University, China, gaged the reactivities of various mononuclear copper-oxygen species in both biological systems and the synthetic systems. The study shows:
These insights are expected to provide the consistent understanding on reactivities of various copper-oxygen active species in both biological and synthetic systems.
The review was published in the Chinese Journal of Catalysis. + Explore further